At Proteowa, we take a collaborative and solutions-focused approach to complex protein expression challenges.
The examples below highlight just a few of the projects we’ve successfully delivered — each reflecting our commitment to technical excellence, innovation, and rigorous quality control.
Note that our services and products are only for scientific research and industrial application, and not available to the public.

Recombinant antibodies
We routinely express monoclonal antibodies and antibody fragments targeting specific antigens, using transient expression in suspension HEK293 or CHO cells. Our process delivers high yield and purity, but what truly sets us apart is the depth of analytical quality control we apply.
At Proteowa, we place a strong emphasis on verifying the structural and functional integrity of every antibody batch. We routinely perform mass spectrometry, HPLC, and electrophoresis to examine molecular weight, glycosylation patterns, and purity. In addition, we use Biolayer Interferometry (BLI) to assess binding affinity and ensure the antibody retains full biological activity.
This thorough approach ensures that clients receive antibodies that are not only pure, but also structurally sound and functionally validated — ready for research or assay development with confidence.
Hemoglobin variants
Human hemoglobin (Hb) is a heterotetramer composed of two α- and two β-chains, each with a heme prosthetic group. Its structural complexity makes heterologous expression challenging, particularly due to limited heme availability and incorporation in E. coli.
Once the protein is harvested, we use a carbon monoxide blanket throughout downstream processing to stabilise the product. Under these conditions, the purified Hb remains stable for weeks at 4 °C or months at –20 °C. Alternatively, it can be stored in stabilising buffers (e.g. ClinRep® Hemolysis Reagent H).
We characterise our Hb products using mass spectrometry, HPLC, UV-visible spectroscopy, and oxygen-binding assays, ensuring structural fidelity and functional performance.

Disulfide-rich proteins
Expressing disulfide-rich proteins in E. coli often results in misfolding due to the reducing nature of the bacterial cytoplasm. Some researchers attempt to address this by using (commercially available) modified strains with altered redox potential; however, these approaches often reduce cell viability and limit expression yield.
At Proteowa, instead of relying on redox-altered strains, we express target proteins as inclusion bodies, then solubilise and refold them in carefully optimised redox environments. This method allows us to achieve high recovery rates of properly folded protein with greater reproducibility and scalability.
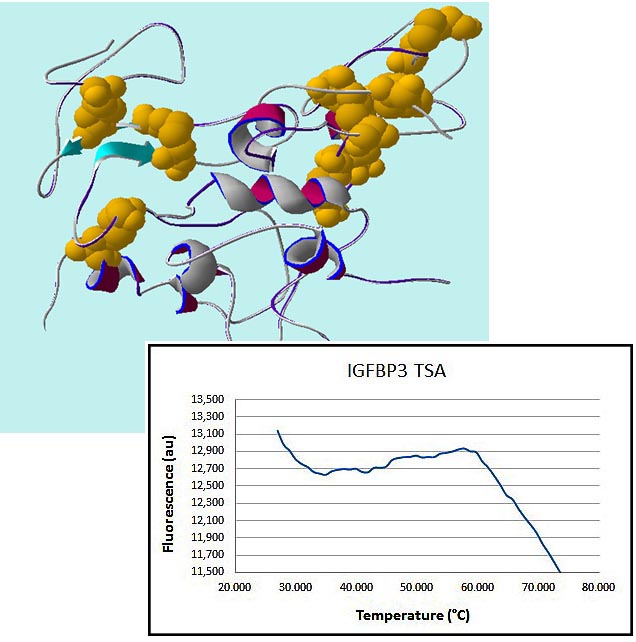
We have successfully applied this strategy to proteins containing multiple disulfide bonds — from typical cytokines and chemokines to complex molecules such as insulin-like growth factor-binding proteins (IGFBPs) containing up to nine disulfide bonds. Folding integrity is confirmed using analytical techniques, and thermal stability is assessed via melting temperature (see an example of the IGFBP3 melting curve on the left).
Chemokines and cytokines
Proteowa has developed robust expression systems for recombinant chemokines and cytokines in E. coli, achieving high yields of biologically active products. We express these proteins as fusion constructs, which are subsequently cleaved to ensure the native N-terminal sequence is retained. Our chemokines and cytokines are verified to have correct sequences, proper disulfide bond formation, and expected biological activities. Purification processes yield products with at least 95% purity, and proteins are sterilized prior to further applications. When required, lyophilization under sterile conditions is performed to facilitate storage and transport.

“Proteowa’s performance-based pricing gave us confidence to pursue a high-risk protein target. Their collaborative and transparent approach made a real difference in moving the project forward.”
Dr Johan M
Scientist
[affiliation witheld]